As the Zika Virus Spreads, Genetically Modified Mosquitoes Become more Real
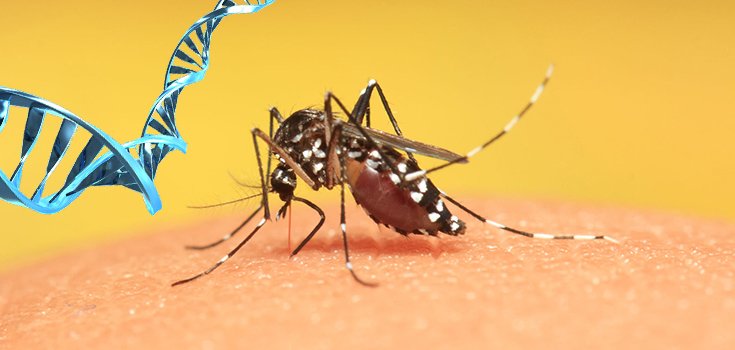
For the average person, the Zika virus is no big deal. Most people never even have symptoms, and those that do have red eyes and feel like the flu. The infection usually clears up within a few days.
In rare cases, Zika can cause Guillain-Barre syndrome, a rare autoimmune disorder that can lead to life-threatening paralysis.
The virus is extremely dangerous for pregnant women, however; Zika has caused thousands of cases of microcephaly in newborns across Brazil. Women are even being told to delay conceiving until 2018 due to the virus.
The virus is in the United States now, though everyone who was been diagnosed here was infected while traveling abroad. But it’s still worrisome, as scientists say it’s only a matter of time until mosquitoes pick up the virus and start spreading it across the U.S.
World leaders are calling on researchers to develop a Zika virus vaccine, but that’s not likely to happen for at least a decade. [1]
To try and bring the spread of the virus under control, Oxitec, a subsidiary of the biotech company Intrexon, is working to create genetically modified mosquitoes – an endeavor the company has been involved with for some time.
Genetically Modified Mosquitoes to the Rescue?
The British firm breeds male mosquitoes that are released into the air and supposedly stop the spread of Zika by passing along a gene to their offspring that makes them die young.
The GM mosquitoes have already been tested in Brazil, Panama, and the Cayman Islands, where researchers claim the Aedes eagypti mosquito population has been reduced by 90% as a result of the GM pests’ release.
Oxitec also claims the GM mosquitoes cut the targeted mosquito population by 82% in Piracicaba, Brazil in a matter of months. The firm says it is expanding the release program in Piracicaba, a city about 100 miles northwest of Sao Paulo. [2]
The firm is awaiting U.S. Food and Drug Administration (FDA) approval to conduct trials of the GM mosquitoes in the Florida Keys. Oxitec and county officials agreed to see if releasing the genetically engineered mosquitoes would reduce the Aedes eagypti population after an outbreak of Dengue fever struck the area several years ago. [3]
The New York Times reported in February, 2015 that the experiments with the genetically modified mosquitoes in the Cayman Islands to fight Dengue showed “limited success.” There was no evidence of any reduction of Dengue. And the article, written by Dr. Helen Wallace, director of GeneWatch U.K., noted computer models showed that it would take the release of 2.8 million GE mosquitoes to reduce a wild population of just 20,000 mosquitoes.
Dr. Wallace specifically lists the very real dangerous posed by releasing GM mosquitoes, including:
“Panamanian researchers have warned that a competitor species, the Asian tiger mosquito, which also transmits dengue and chikungunya, could move in and be harder to eradicate. Disease transmission by this species might increase in the future.
Researchers in Germany have highlighted that some biting female genetically engineered mosquitoes will be released, and that Oxitec has not provided sufficient evidence that been bitten by, or swallowing, these mosquitoes will be safe. (Oxitec says it only releases male mosquitoes, but inevitably some females do get released.)
Oxitec uses tetracycline as a chemical switch for the genetic killing mechanism in its genetically engineered mosquitoes. Survival rates of next-generation genetically engineered mosquitoes increase from 3 percent up to 18 percent when fed on industrially farmed meat, which is contaminated with the common antibiotic tetracycline. Tetracycline will be present in release areas in discarded takeaways, pet food and in some mosquito breeding sites such as septic tanks.
The use of tetracycline to feed genetically engineered mosquitoes in Oxitec’s mosquito factory risks spreading antibiotic resistant bacteria into the environment, posing a risk to human health. The non-veterinary use of tetracycline is being phased out in the United States and is already banned in many countries.
The regulatory process being followed by the Food and Drug Administration remains unclear. Company press releases should not replace the need for detailed evidence and public consultation on the risk assessment.” [4]
Dr. Joseph Mercola notes on his website:
“- The potential exists for these genes, which hop from one place to another, to infect human blood by finding entry through skin lesions or inhaled dust. Such transmission could potentially wreak havoc with the human genome by creating “insertion mutations” and other unpredictable types of DNA damage.
– According to Alfred Handler, a geneticist at the Agriculture Department in Hawaii, mosquitoes can develop resistance to the lethal gene and might then be released inadvertently.
Todd Shelly, an entomologist for the Agriculture Department in Hawaii, said 3.5 percent of the insects in a laboratory test survived to adulthood, despite presumably carrying the lethal gene.” [5]
Oxitec also has a history of conducting its experiments in secret. Key West, Florida, residents were outraged to learn the firm was working alongside the Florida Keys Mosquito Control District, without consulting them and without conducting studies on their methods.
“I’m all for the experiment, but it has to be safe. Why can’t we have independent, peer-reviewed research to prove it?” one Key West resident asked.
Oxitec was criticized by organizations worldwide when it released 3.3 million GM mosquitoes in the Cayman Islands in 2009 without consulting the public, and without independent oversight. The biotech company went ahead and expanded its operations to Malaysia and Brazil, despite protests. [6]
So far, Oxitec has only gained regulatory approval to release the mosquitoes in Brazil. The mosquitoes are only being approved in other countries on a case-by-case basis.
Sources:
[1] Daily Mail
[2] CNN Money
[3] NPR
[5] Mercola

To start with, there were NEVER an epidemic of dengue in the Cayman Islands.
“It’s a miracle – How Jaxon, the baby given days to live when he was born with most of his skull missing, has defied the odds – and is celebrating his first birthday. DailyMail September 26,2015”. Was Zika in FL already?
From one of the cross-links – Oxitec is quickly trying to remove itself from Any harm caused by the unnatural GM mosquitoes, to wit:
Doesn’t want to be liable for any complications.
Tries to avoid any regulation of GM agricultural pests on crops appearing in the food chain.
Excludes important issues from risk assessments, such as the impact
on human immunity and disease, and the possible outcomes arising from
surviving GM mosquitoes.
Releases large amount of GM mosquitoes prior to regulations.
Attempts to define ‘biological containment’ of the insects (which
are programmed to die at the larval stage) as contained use, by-passing
requirements for risk assessments and consultation on decisions to
release GM insects into the environment.
Undermining the requirement to obtain informed consent for experiments involving insect species which transmit disease.
Ignores any labeling using products produced from GM insects and how insects can be contained where released.
Very dangerous experiment on the human population. Oxitec had been releasing GM mosquitoes in Brazil since 2011. .3% females released and 15% offspring survive and unknown number get tetracycline. They’re made with E.coli and Herpes virus, its no wonder the newer Zika virus has been developed as feared a worse strain of it could happen.