Scientists Create Human-Pig Embryo for Transplant Research
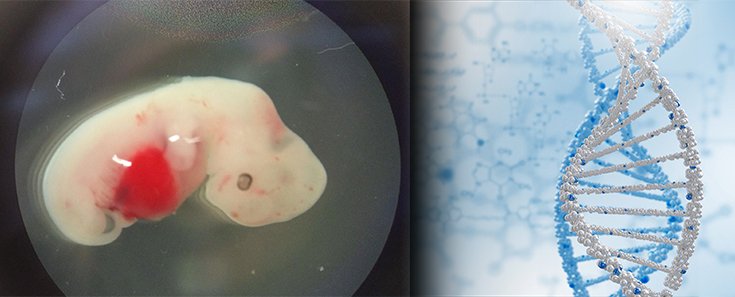
Scientists have created embryos that are part-pig, part-human for the purpose of studying how to grow organs for human transplant. The experiment is detailed in the journal Cell. [1]
Researchers injected human stem cells into a pig embryo and then implanted the embryo into the womb of a sow and let it grow. Four weeks later, the stem cells had developed into the beginnings of various types of tissue, including heart, liver, and neurons. A small fraction of the developing pig consisted of human cells.
In the tissue that would go on to become the embryonic pig’s heart, 1 of every 1,000 to 10,000 cells was human. In the embryo as a whole, human cells accounted for approximately 1 in every 100,000 cells. Human cells had not made their way into the pigs’ brain or gonadal tissue, but some of them were found in peripheral nervous system tissue. Future research will focus on how to increase the concentration of human cells in certain organs, like the heart. [2]
Human-pig embryos have been created by scientists before. Over the summer, researchers announced that they’d implanted human-pig embryos into sows, terminating the pregnancies after 28 days. For that experiment, researchers used CRISPR gene-editing technology to remove DNA from the embryos that would allow the subsequent fetus to grow a pancreas, creating a genetic “niche.” Then human induced pluripotent stem (iPS) cells were injected into the embryo. Researchers “dialed back” the iPS cells so that they would become stem cells that are programmable to develop into any tissue in the body.
Read: Scientists Successfully Grow Living Organ from Stem Cells for First Time
News of the most recent human-pig hybrids broke just a day after researchers demonstrated that organs for transplant can be grown in chimeras that are part-mouse, part-rat.

In that case, scientists were able to grow a mouse pancreas inside of a rat embryo, then transfer insulin-secreting tissue from the pancreas into diabetic mice, reversing the disease and keeping the rodents’ sugar levels in check for a year.
CRISPR gene-editing was also used in the mouse-rat chimeras so that the rats would not grow their own pancreases.
Both scientific advances present a mind-bending ethical conundrum. In 2015, the National Institutes of Health (NIH) ordered a moratorium on funding for certain human-animal chimera research, though they have recently proposed loosening some of those limits. (The new study was conducted at the Salk Institute in California without federal funds.) Stem cells have the ability to become any type of cell in the body, raising the possibility of horrific combinations of animals with a human brain or reproductive organs. [1] [2]
For example, if some of the stem cells entered the gonadal tissues of pigs, profound genetic changes could be introduced that would be passed on for generations. If that’s not scary enough for you, imagine a pig with the consciousness or intellectual ability of a human – even a fraction of it.
Carrie D. Wolinetz, the institutes’ associate director for science policy, explained:
“There has been the use of human-animal chimeras in biomedical research for a very long time. We’ve introduced human cells into animal models to create models of human diseases and used them as research tools for decades now.
But over time, what we’ve seen — as stem cell technology has advanced and as gene editing technology has advanced — we’ve seen the ability to create more sophisticated animal-human chimeras at a much earlier stage of embryonic development.” [3]
Arizona State University bioethicist Jason Robert said that in the future, researchers might introduce human cells into the embryonic tissue of higher-order animals like primates. [2]
Dr. Jun Wu, a staff scientist in the gene expression laboratory at the Salk Institute and first author of the research, said:
“Species evolve independently, and many factors dictating the developmental programs might have diverged, which makes it difficult to blend cells from one species to a developing embryo from another. The larger the evolutionary distance, the more difficult for them to mix.” [3]
It’s unlikely that using primates instead of pigs would alleviate ethical concerns, however.
Sources:
[3] CNN
Technology Review (Featured image photograph courtesy Juan Carlos Izpisua Belmonte)
