U.S. Regulators Warn Some Teething Medicines Are Unsafe for Babies
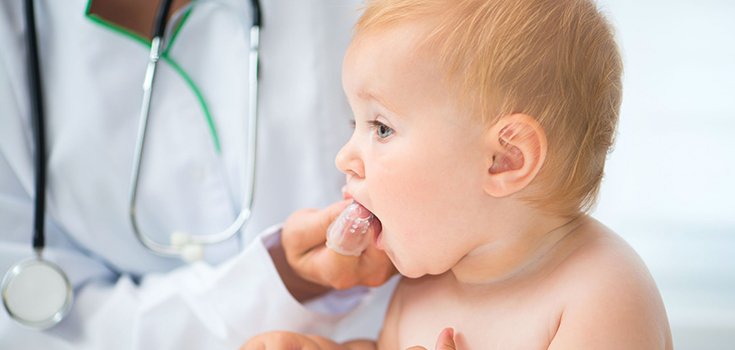
The U.S. Food and Drug Administration (FDA) warned earlier this year that some teething medicines are unsafe for babies and asked manufacturers to stop selling their products intended for babies and toddlers.
Teething gels and creams made with benzocaine, a numbing agent, can cause rare but deadly side effects, particularly in children 2 years and younger, according to the agency.
Some of the medicines on the chopping block include Orajel and Anbesol as well as generic drugstore brands. These benzocaine-containing products are used to soothe toothaches and sore gums. Products for adults can stay on the market, but the FDA wants companies to add new warnings.
Benzocaine can cause a rare blood condition known to lead to potentially deadly breathing problems. The ingredient can interfere with a protein that carries oxygen through the blood, resulting in symptoms such as shortness of breath, headache, and rapid heart rate.
In a statement, FDA Commissioner Scott Gottlieb said:
“We urge parents, caregivers, and retailers who sell them to heed our warnings and not use over-the-counter products containing benzocaine for teething pain.”
Church and Dwight Co. said it would stop manufacturing 4 Orajel teething brands, including Orajel Medicated Teething Swabs.
The company said in a statement:
“We are not discontinuing other Orajel products, which represent the majority of our Orajel offerings.”
Teething creams are frowned upon by the American Academy of Pediatrics, as the products tend to wash out of the baby’s mouth minutes after being applied. The group recommends giving babies a teething ring or massaging their gums, instead.
This is not the first time the FDA has issued a warning about teething products. The agency raised the alarm in 2006, 2011, and 2014 but stopped short of removing the medicines from the market. The latest warning was triggered by a review of 119 cases of the benzocaine-linked breathing disorder between 2009 and 2017, including 4 deaths, the regulator said.
