Infant Liquid Ibuprofen Recalled at Walmart, CVS, Other Retailers
Cold and flu season is upon us, so the medicines-intake will likely be rolling around. But if you’ve recently purchased liquid ibuprofen from Walmart, CVS, or Family Dollar, you should stop using it immediately and return it for a refund. The over-the-counter pain relievers were recalled December 6 because they may contain more ibuprofen than what is listed on the label.

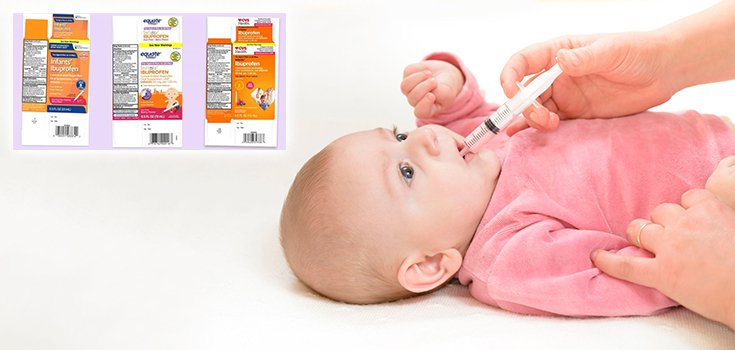
The recalled fever-reducing and pain-relieving products were packaged and sold as follows:
- At Walmart stores as Equate: Infants’ Ibuprofen Concentrated Oral Suspension, USP (NSAID), 50 mg per 1.25 mL, 0.5 oz. bottle. Expiration 02/19, lot 00717009A; expiration 04/19, lot 00717015A; expiration 08/19, lot 00717024A. NDC (national drug code) 49035-125-23.
- At CVS Pharmacies as CVS Health: Infants’ Ibuprofen Concentrated Oral Suspension, USP (NSAID), 50 mg per 1.25 mL, 0.5 oz. bottle. Expiration 08/19, lot 00717024A. NDC 59779-925-23.
- At Family Dollar Stores as Family Wellness: Infants’ Ibuprofen Concentrated Oral Suspension, USP (NSAID), 50 mg per 1.25 mL, 0.5 oz. bottle. Expiration 08/19, lot 00717024A. NDC 55319-250-23.
The higher concentrations of ibuprofen in the recalled products put infants at a slightly increased risk of kidney damage. The excess medication may also cause:
- Nausea
- Vomiting
- Upper abdominal pain
- Ringing in the ears (tinnitus)
- headache
- Intestinal bleeding are also possible
- Less commonly, diarrhea
Ibuprofen belongs to a family of medicines known as nonsteroidal anti-inflammatory drugs (NSAIDs) that are associated with stomach bleeding.
Fortunately, Tri Pharma has not received any reports of adverse events caused by the affected products.
Parents and caregivers with questions can contact Tri Pharma via phone at 732-940-0358, Monday through Friday, 8 a.m. to 5 p.m. ET.
Stop using the medications in question immediately. Stores typically refund recalled products.
Furthermore, if your child experiences any problems as a result of taking the recalled infant ibuprofen, you should contact your doctor and report your experiences to the U.S. Food and Drug Administration’s (FDA) MedWatch Adverse Event Reporting program. You can either contact the program online, or download a form and return it by mail at the address on the form, or fax it to 800-FDA-0178 (800-332-0178).
Source:
[1] WebMD
